Introduction
Male reproductive function depends on the integrity of the organs of the male reproductive tract, most relevantly, the testis, epididymis, deferent duct, seminal vesicle, prostate, and penis, as well as the various parts of the central nervous system, the hypothalamus, and the pituitary gland. In contrast to female reproductive function, hormone and gamete production in healthy men persists lifelong. However, various disorders of male reproductive function show a relatively high prevalence and are therefore of significant relevance for public health.
The Testis
The main male reproductive organ is the testis. The testis has an endocrine as well as an exocrine function. The predominant endocrine function is synthesis of testosterone, which is important for normal sperm production within the testis as well as – after entering the general circulation and binding to the androgen receptors of the various target organs – for initiating and maintaining androgen-dependent functions. The most important exocrine function of the testis is production of immature spermatozoa that are – after maturation in the epididymis – able to fertilize an egg and induce a pregnancy.
Endocrine testicular dysfunction is characterized by testosterone deficiency. A recent population-based, observational survey indicates that the overall prevalence of testosterone deficiency associated with relevant clinical symptoms in men aged 30–79 years is 5.6% (Araujo et al., 2007). These data demonstrate the overt relevance of testosterone deficiency for public health. In primary endocrine testicular failure, low testosterone secretion is caused by a deficiency or absence of Leydig cell function. Clinically relevant diseases include anorchia, gonadal dysgenesis, testicular tumors, testicular maldescent, Leydig cell hypoplasia, enzymatic defects in testosterone synthesis, numerical or structural chromosome abnormalities, gene mutations, irradiation, and systemic diseases. Testosterone deficiency can also be caused by exogenous factors such as environmental toxins or medications (Table 1). In contrast to primary endocrine testicular failure, secondary endocrine testicular failure is caused by absent or insufficient bioactivity of gonadotropin-releasing hormone (GnRH) or luteinizing hormone (LH).

Men diagnosed with primary exocrine testicular dysfunction are infertile. Although exact figures are not available, it is assumed that about 7% of all men are confronted with impaired fertility in the course of their lives, resulting in a significant impact on public health (Nieschlag, 2000b). Infertility might be caused by impaired spermatogenesis due to deficiency of GnRH or gonadotropin secretion, anorchia, gonadal dysgenesis, varicocele, orchitis, structural abnormalities of spermatozoa, testicular tumors, testicular maldescent, Leydig cell hypoplasia, enzymatic defects in testosterone synthesis, irradiation, or systemic diseases, or by exogenous factors such as environmental toxins or medications, numerical chromosome abnormalities, structural chromosomal abnormalities, Y chromosome microdeletions, gene mutations, or other still unknown genetic defects (Table 1).
Hormonal Regulation Of Testicular Function
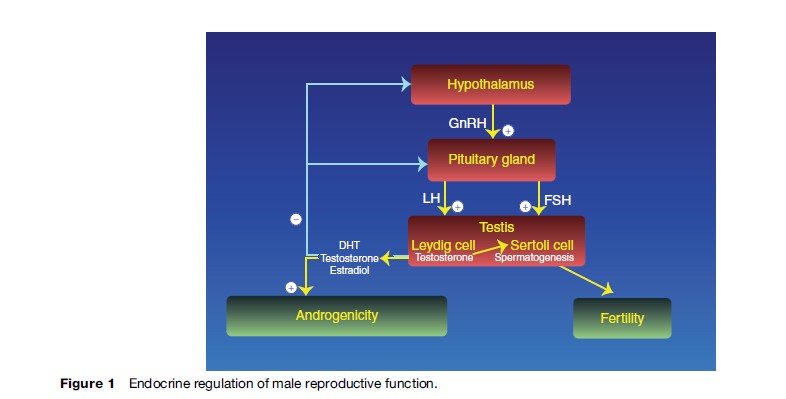
Testicular steroidogenesis and spermatogenesis are controlled primarily by the hypothalamic GnRH and the two pituitary gonadotropins, luteinizing hormone and folliclestimulating hormone (FSH) (Figure 1). The production and secretion of these stimulating hormones are under negative feedback control of hormones produced by the testis, especially testosterone and its active metabolites and inhibin B (Weinbauer et al., 2000).
Gonadotropin-Releasing Hormone (Gnrh)
GnRH is a decapeptide produced in specific neurons of the hypothalamus. It has a relatively short half-life of less than 10 min and is released into the portal blood in discrete pulses, which are at least partially controlled by stimulatory and inhibitory effects of various steroid hormones, neurotransmitters, and neuromodulators. GnRH stimulates LH and FSH synthesis and secretion from the pituitary gland. The nature of the pulsatile GnRH secretion has not been well-characterized in the human but seems to be important for GnRH action, as experimental long-term continuous GnRH release is not able to stimulate LH and FSH synthesis and secretion.
GnRH stimulates gonadotropin secretion following occupation and activation of specific GnRH receptors in the pituitary gland. These receptors belong to a family of G protein-coupled receptors having the typical sevenmembrane domain structure. GnRH is capable of modulating the number and activity of its own receptor.
Several disorders and diseases are known to cause testicular dysfunction by disturbances of GnRH synthesis, secretion, or function (Table 1). Diminished or absent hypothalamic secretion of GnRH can be found in patients with idiopathic hypogonadotropic hypogonadism (IHH) or Kallmann syndrome. Secondary GnRH deficiency might be caused by a tumor in the region of the diencephalon (craniopharyngioma or meningioma), metastases, granulomatous illnesses or hemochromatosis, fractures of the skull base, ischemic and hemorrhagic lesions, or radiotherapy. In patients with absent or diminished GnRH secretion, highly effective restoration of testicular function can be achieved by exogenous administration of GnRH intravenously or subcutaneously, in a pulsatile manner (Buchter et al., 1999).
Several patients with inactivating mutations of the GnRH receptor causing testicular dysfunction have been described. In these men, exogenous GnRH is ineffective, but proper treatment of infertility can be achieved by exogenous gonadotropin administration (see ‘Gonadotropins’). If fertility is not desired, patients should be treated with testosterone preparations.
Gonadotropins
LH and FSH, the gonadotropins regulating testicular function, are glycoprotein hormones secreted from the pituitary gland in response to GnRH (Figure 1). They consist of two subunits, alpha and beta. While the alpha subunit of LH and FSH is identical and is similar to the alpha subunit of other members of the glycoprotein hormone family, such as thyroid stimulating hormone or human chorionic gonadotropin (hCG), the specific actions of LH and FSH are caused by their specific beta subunits. Due to different terminal glycosylation, LH and FSH have different elimination half-lives of about 20 min and 120 min, respectively. Therefore, the pulsatility as measured in serum is more apparent for LH than for FSH. In addition to the pulsatile secretion of both gonadotropins, FSH is, to some degree, secreted constitutively from the pituitary gland.
Steroid hormones secreted from the testes, especially testosterone and its active metabolites, dihydrotestosterone and estradiol, exert a negative feedback control of both gonadotropins at the level of both the hypothalamus and pituitary. In addition, FSH secretion is under negative control of inhibin B secreted from the Sertoli cells of the seminiferous tubules in the testes.
Both gonadotropins act via specific receptors on different testicular cell types, with LH exerting its effects on Leydig cells and FSH binding to and activating receptors in Sertoli cells. The LH and FSH receptors belong to the family of G protein-coupled receptors, and both have a large extracellular hormone-binding domain. Normal function of both gonadotropins and their receptors seem to be necessary for quantitatively and qualitatively normal initiation and development of spermatogenesis at puberty. Although some degree of spermatogenesis might be maintained thereafter by one normally functioning gonadotropin and its receptor, both LH and FSH activity seem to be mandatory for normal spermatogenesis.
A lack of normal physiological levels of gonadotropins in patients might be caused by tumors (gonadotropin, prolactin, growth hormone, thyroid hormone, ACTH-secreting, or endocrinologically inactive adenomas) or metastases of tumors in the pituitary or the hypophyseal stalk, brain surgery, radiotherapy, trauma, infections, hemochromatosis, or vascular disorders (Table 1). The most frequent cause of pituitary insufficiency is a prolactin-secreting pituitary adenoma (prolactinoma) that might destroy the gonadotropinsecreting pituitary cells or impair their function.
In most patients, prolactinomas can be treated effectively with dopamine agonistic drugs such as bromocriptine. Patients with irreversible destruction of gonadotropinsecreting pituitary cells can be successfully treated for their infertility with administration of exogenous gonadotropins. Instead of the natural LH, the gonadotropin hCG – which has LH activity – is commonly used because of its prolonged duration of action (Bu¨ chter et al., 1999).
In some patients, inactivating mutations of the LH or FSH receptor gene have been detected as the cause for testicular dysfunction. In these patients, gonadotropin preparations with LH and FSH activity are generally not effective.
Endocrine Function: Testosterone Production
Testosterone is the most important sex hormone in men. More than 95% of the endogenous testosterone is produced in the testes, with 6–7 mg secreted every day. Testosterone is produced by the Leydig cells in the interstitial compartment in response to LH binding to its specific Leydig cell membrane receptor. As no testosterone can be stored in the Leydig cells, it has to be produced continuously de novo.
Testosterone is an important intratesticular factor for regulating spermatogenesis. In addition, it is released into the general circulation and transported in serum, with 54% bound to sex hormone binding globulin (SHBG), 44% bound to albumin, and 2% circulating as free or unbound steroid. The free and albumin-bound fractions are considered to be biologically active. However, the physiological role of SHBG-bound testosterone has not yet been fully elucidated. Several studies have shown that the testosterone concentration within the testis is about 100-fold higher than in the general circulation.
In serum, testosterone levels follow a circadian pattern, with morning concentrations 20–40% higher than evening values (Diver et al., 2003). As normal ranges for serum testosterone levels are commonly based on morning levels, blood for diagnostic tests should be drawn in the morning. Short, intensive physical exercise can increase serum testosterone concentrations, whereas extended, exhausting physical exercise and the practice of high-performance sports can result in significant decreases.
Testosterone has two clinically relevant metabolites. It is 5 alpha-reduced to 5a-dihydrotestosterone (DHT), which is important for normal development of the external male genitalia and prostate gland. In a separate metabolic pathway, testosterone is aromatized to 17b-estradiol, which has essential functions in bone development.
Testosterone and its active metabolite DHT bind to an intracellular androgen receptor that belongs to the family of steroid hormone receptors. Once occupied, these receptors translocate to the cell’s nucleus and bind to specific sequences of the genomic DNA to induce RNA and protein synthesis. In addition, a rapid nontranscriptional mode of action has been recently demonstrated for testosterone and DHT.
Various androgen receptor mutations have been described in infertile patients; these result in diminished or absent androgen action. In addition, polymorphisms of the androgen receptor, especially the so-called CAG repeat polymorphism in exon 1 of the androgen-receptor gene, have been shown to modify androgen action (Zitzmann and Nieschlag, 2007). In men with higher numbers of triplet residues, testosterone’s effects in the various target organs seem to be attenuated.
Hypogonadism
Hypogonadism can be defined as a condition with absent or decreased biological action of testosterone. It might be caused by disorders at the testicular level and is then classified as primary hypogonadism. Hypogonadism due to disorders at the hypothalamic or pituitary level is classified as secondary hypogonadism. In addition, hypogonadism might be caused by androgen receptor dysfunction at the androgen target organs (Table 1).
The clinical symptoms of hypogonadism depend on the age of manifestation, in particular whether onset occurs before completed puberty or thereafter (Table 2). If androgen deficiency exists at the time of normal onset of puberty, an eunuchoid tall stature will result because of delayed epiphyseal closure. Arm span will exceed body length and the legs will become longer than the trunk. Onset of androgen deficiency after puberty will not result in a change of body proportions, although the musculature can be atrophic, depending on the duration and degree of androgen deficiency.
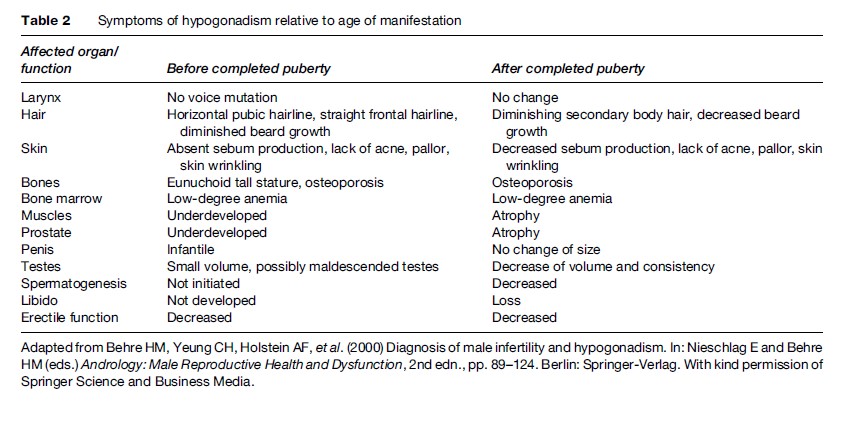
Long-standing testosterone deficiency may lead to osteoporosis, which can result in severe lumbago and pathological spine and hip fractures. Fat tissue distribution might have female characteristics, and lean body mass can be decreased and fat mass increased compared to normal.
Late-onset hypogonadism (LOH), defined as testosterone deficiency with clinical symptoms in the aging male, has become a topic of increasing interest throughout the world (Nieschlag et al., 2005). Current demographic trends show a worldwide ‘graying’ of the population, with increasing percentage of the population aged 60–65 years and above. Average testosterone levels fall progressively with age, and a significant percentage of elderly men have serum testosterone levels lower than the normal ranges of young adults. A recent study estimated the overall prevalence of testosterone deficiency in combination with typical clinical symptoms of hypogonadism to be as high as 5.6% in men aged between 30 and 79 years. Prevalence was not related to race or ethnic group; however, it increased substantially with increasing age (Araujo et al., 2007).
Late-onset hypogonadism is often caused by mixed primary and secondary endocrine testicular failure. Symptoms of LOH are given in Table 3. However, the clinical diagnosis might escape detection for various reasons: Not all the signs and symptoms necessarily present together, they often progress slowly and are subtle in nature, and the unspecific signs and symptoms of LOH might not be discernible from the unavoidable process of aging itself (Nieschlag et al., 2004).

The most comprehensive review of the long-term clinical consequences and morbidity of LOH has been compiled by the U.S. Institute of Medicine (Liverman and Blazer, 2004). Potential impacts of age-related testosterone decline on health have been identified for bone, body composition and strength, physical function, cognitive function, mood and depression, sexual function, health-related quality of life, cardiovascular and hematologic outcomes, prostate outcomes, sleep apnea, and other areas.
Apparent good health, defined as the absence of chronic illness, medication, obesity, or excessive drinking, attenuates the age-related decrease of testosterone (Feldman et al., 2002). On the other hand, at all ages, serum testosterone concentrations may be transiently or permanently affected by comorbidities or their respective treatment (Kaufman and Vermeulen, 2005). Recently, it has been demonstrated in men aged 45 years and visiting primary care practices in the United States that odds ratios for having hypogonadism were significantly higher for men with hypertension, hyperlipidemia, diabetes, obesity, prostate disease, and asthma or chronic obstructive pulmonary disease than in men without these conditions (Mulligan et al., 2006). Therefore, a proper diagnostic workup is mandatory in all men with known or suspected testosterone deficiency.
In patients with disturbed endocrine testicular function and without present intention to induce a pregnancy, the therapy of choice is testosterone substitution. Numerous studies and more than 50 years of clinical experience have confirmed that testosterone substitution has beneficial effects on the symptoms of patients with primary or secondary hypogonadism, with acceptable adverse side effects. Today, several testosterone preparations are available for clinical use that can be applied via the intramuscular, transdermal, subcutaneous, oral, or buccal route. The various preparations have different pharmacokinetics, different adverse side effects, and different costs. As evidence from comparative studies is not available, the choice of the testosterone preparation is based on personal preferences and experience of the patient and the treating physician.
Men with late-onset hypogonadism may or may not benefit from testosterone treatment; the risks associated with such intervention are not well-described for this population (Nieschlag et al., 2005). Results from recent studies show short-term beneficial effects of testosterone in older men that are similar to those in young men, but long-term data on the effects of such treatment in men with late-onset hypogonadism are limited. Future studies have to evaluate the specific risks on the prostate and cardiovascular system as well as the potential long-term benefits, including those that may retard frailty of elderly men.
Exocrine Function: Spermatogenesis
The second major function of the testis is production of spermatozoa. This process, termed spermatogenesis, takes place in the tubular compartment of the testis, while the testosterone production of the Leydig cells is located in the interstitial compartment (Weinbauer et al. 2000).
The tubular compartment of the testis represents 60–80% of the testicular volume. In total, each testis contains about 600 seminiferous tubules. The Sertoli cells within the seminiferous tubules are somatic cells and extend from the basal membrane to the lumen of the tubule. Sertoli cells support the structure of the semi- niferous tubule and have a role in regulating the internal environment. The number of Sertoli cells in a testis determines the ultimate spermatogenic output. Sertoli cell function is regulated by occupation and activation of androgen and FSH receptors, and these hormonal signals are then transduced to the germinal cells.
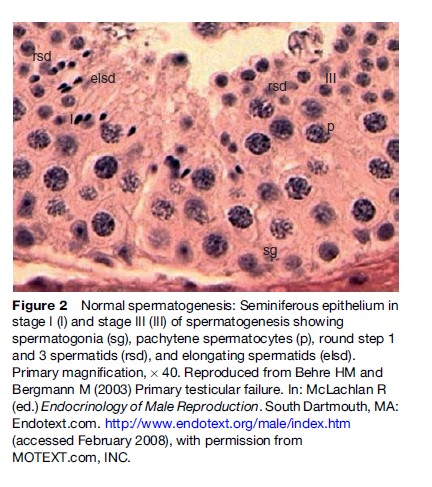
Spermatogenesis takes place in three phases: (1) mitotic proliferation and differentiation of spermatogonia, (2) meiotic division, and (3) spermiogenesis, which represents a transformation of round haploid spermatids arising from the final division of meiosis into the complex structure of the spermatozoon (Figure 2). The overall duration of spermatogenesis is at least 64 days.
In most men with severely reduced sperm concentration in the ejaculate and no obstruction of the seminal ducts, various disturbances of spermatogenesis can be detected by testicular biopsy. In complete germ cell aplasia or Sertoli-cell-only syndrome (SCO), the tubules are reduced in diameter and contain Sertoli cells, but no other cells involved in spermatogenesis. Germ cell aplasia can also be focal with a variable percentage of tubules containing germ cells, but in these tubules spermatogenesis is often limited. Germ cell aplasia or SCO syndrome is one common cause of nonobstructive azoospermia (complete absence of spermatozoa in the ejaculate).
Spermatogenic arrest is a histopathological description of the interruption of normal germ cell maturation at the level of a specific cell type in the pathway leading from spermatogonia to spermatocytes to spermatids. Complete arrest of spermatogenesis results in azoospermia. Spermatogenetic arrest or germ cell aplasia might be caused by, for example, microdeletions of the Y chromosome and numerical or structural chromosome abnormalities.
Hypospermatogenesis is the third major classification of impaired spermatogenesis; it is characterized by a mildly, moderately, or severely reduced number of all germ cell types, including mature spermatids, in some or all tubules.
Seminal Ducts
Epididymis
After production within the seminiferous tubules of the testis, the immature testicular spermatozoa together with fluid secreted by the Sertoli cells pass the tubuli recti and the rete testis and enter into 12–18 efferent ducts that form the caput of the epididymis. Here, the efferent ducts unite and form the single tubule of the epididymis. The single tubule of the epididymis is then coiled into lobules and forms the corpus epididymis and finally the cauda epididymis. The total length of the epididymis is estimated to be 5–6 m. The time for spermatozoa to pass through the epididymis is between 2 to 11 days. The major function of the epididymis is the maturation of spermatozoa and storage prior to ejaculation.
Deferent Duct And Seminal Vesicle
During the emission phase of ejaculation, spermatozoa from the lower portion of the epididymis are transported via the deferent duct through its peristaltic action of muscle layers to the intraprostatic ejaculatory duct. Each deferent duct is about 30 cm long, and the ejaculatory duct about 2 cm. The seminal vesicle is a tubular gland of approximately 5 cm in length, located posteroinferior to the urinary bladder and surrounded by a thick coat of smooth muscle. The capacity of each seminal vesicle is approximately 2 ml, which contributes about 70% of the ejaculate volume. During the emission phase of ejaculation, sperm-containing fluid of the proximal deferent duct is diluted with fluids of the seminal vesicle, and the mixture transported via the intraprostatic ejaculatory duct into the prostatic urethra at the seminal colliculus.
Prostate
The prostate is a tubulo-alveolar exocrine gland surrounding the urethra just below the urinary bladder. Its main function is storage and secretion of clear alkaline fluid that constitutes up to 30% of the ejaculate volume. The prostatic urethra transports urine or, during ejaculation, the seminal fluid containing spermatozoa. During the ejection phase of ejaculation, the bladder neck becomes firmly closed, preventing reflux of the ejaculate into the bladder. By contraction of the prostatic musculature and pelvic floor muscles, the ejaculate is propelled into the anterior penile urethra and beyond. Ejaculatory disorders include retrograde, premature, retarded, or absent ejaculation, as well as various orgasmic disturbances. There are no good data on the prevalence of ejaculatory disorders; however, they seem to be as frequent as erectile dysfunction. For example, premature ejaculation has been found to affect approximately 13–18% of men aged between 40 and 80 years (Beutel et al., 2006).
Penis
From the male reproductive function perspective, normal erectile function of the penis is important for sexual intercourse resulting in proper intravaginal sperm deposition for successful reproduction. Three hemodynamic factors in the penis are essential for normal erection: (1) relaxation of the muscle cells within the corpora cavernosa of the penis, (2) increase of arterial blood inflow by dilatation of the arterial vessels, and (3) restriction of venous blood outflow by compression of intracavernosal and subtunical venous plexus.
According to the National Institutes of Health consensus, erectile dysfunction is defined as the inability to achieve and maintain an erection sufficient for satisfactory sexual performance. Erectile dysfunction can be caused by various disturbances. It is estimated that 50–80% of organically caused erectile dysfunction is due to arterial dysfunction. Other common reasons include neurogenic, endocrine, drug-induced, and – more often in younger men – psychogenic etiologies. In many patients, the reason for erectile dysfunction is multifactorial. Numerous epidemiological studies have been published on the prevalence of erectile dysfunction. In all these studies, the prevalence was strictly associated with aging. The overall prevalence has been estimated to be as low as 2.3% in men aged 30–39 years, but up to about 15–70% in men aged above 70 years (Beutel et al., 2006).
Infertility
Infertility may be diagnosed when a couple fails to achieve a pregnancy within 1 year of regular unprotected intercourse. In contrast to other diseases or disorders, the diagnosis of infertility can be made only in a couple, and only in a couple with a desire for pregnancy. There is a significant interdependence of the reproductive function of the male and female partners. Disturbed male reproductive function, such as decreased sperm count, can be compensated for by optimal reproductive functions of the female partner, and vice versa.
Various diseases in the male might cause disorders of male reproductive function and infertility. These conditions can be classified by the topographic localization of the cause (Table 1). There are several causes for primary and secondary testicular failure. In many patients who have various degrees of abnormal spermatogenesis, the exact cause for testicular dysfunction is unknown. These patients are categorized as having idiopathic infertility and remain a majority of patients seen in modern andrology practices. For these patients, symptomatic treatment applying methods of assisted reproduction can be offered, such as intrauterine insemination, intracytoplasmic sperm injection with ejaculated sperm or sperm retrieved from testicular biopsies by testicular sperm extraction (TESE), depending on the site and degree of spermatogenic failure.
Acute or chronic infections of the epididymis, seminal vesicles, or prostate are curable conditions of male infertility. In patients with bilateral obstruction of the epididymis or deferent duct, microsurgical reanastomosis can be offered to restore fertility. When this is not possible, for instance in patients with congenital bilateral aplasia of the vas deferens/deferent duct (CBAVD), sperm can be retrieved from the epididymis by microsurgical sperm aspiration, or from testicular biopsies by TESE. For patients with CBAVD, a condition caused by mutations in the cystic fibrosis transmembrane conductance regulator gene, genetic counseling and testing should be offered.
In couples unable to conceive due to erectile dysfunction, various highly effective therapies can be offered, such as phosphodiesterase type V inhibitors, intracavernosal prostaglandin E1 injections, and hormonal, surgical, or psychological therapy. After proper diagnosis of the etiology of erectile dysfunction in the individual affected patient, the effectiveness of these rational therapies is high.
Infertility can be caused by retrograde ejaculation, for example, after surgery or trauma. In affected patients, the ejaculate – which would normally exit via the urethra – is redirected toward the urinary bladder. Patients with retrograde ejaculation can effectively be treated with various drugs, such as imipramine or chlorpheniramine in combination with phenylpropanalamine, which produce antegrade ejaculation and thereby restore fertility.
The multiplicity of diseases, disorders, or exposures resulting in infertility clearly indicates the need for a systematic andrological examination of the affected man, and not just an analysis of his ejaculate. Rational and preventive therapy of male infertility is based on proper clinical and laboratory diagnoses. These should include a comprehensive history, physical examination, application of ancillary methods such as ultrasonography of the testes (Figure 3), prostate, and seminal glands, and proper endocrine laboratory diagnosis and semen analysis, according to standard procedures recommended by the World Health Organization (World Health Organization, 2008). In certain patients, examination might be completed by testicular biopsies and molecular and cytogenetic analyses.
Whenever possible, preventive therapy should be applied to maintain normal male reproductive function and integrity of the reproductive organs. For example, timely use of effective antibiotics in case of infections of the epididymis or prostate might prevent irreversible obstruction leading to azoospermia. Early hormonal treatment of maldescended testes or, if unsuccessful, surgical therapy might preserve fertility and decrease the risk of future testicular cancer (Pettersson et al., 2007). Exogenous factors, such as radiation or environmental toxins, should be minimized or eliminated as soon as possible. Prior to compulsory chemotherapy or radiotherapy because of malignant diseases, patients should be offered cryopreservation of spermatozoa in order to maintain reproductive ability. From the public health perspective, it should be noted that smoking, obesity, abuse of alcohol or certain drugs such as anabolic steroids, and untreated systemic diseases are potentially avoidable factors leading to infertility (Handelsman, 2000; Brinkworth and Handelsman, 2000).
Conclusion
Male reproductive function depends on normal functioning of the various organs of the male reproductive tract and the respective regulatory mechanisms of the central nervous system. It should be noted that disturbances of reproductive function, such as hypogonadism, ejaculatory disorders, erectile dysfunction, and infertility, have a relatively high worldwide prevalence of more than 5% for each disorder. Therefore, proper diagnosis and treatment of male reproductive dysfunction as well as prevention whenever possible are of utmost relevance for public health.
Bibliography:
- Araujo AB, Esche GR, Kupelian V, et al. (2007) Prevalence of symptomatic androgen deficiency in men. Journal of Clinical Endocrinology and Metabolism 92: 4241–4247.
- Behre HM and Bergmann M (2003) Primary testicular failure. In: McLachlan R (ed.) Endocrinology of Male Reproduction. South Dartmouth, MA: Endotext.com. http://www.endotext.org/male/ index.htm (accessed February 2008).
- Behre HM, Yeung CH, Holstein AF, et al. (2000) Diagnosis of male infertility and hypogonadism. In: Nieschlag E and Behre HM (eds.) Andrology: Male Reproductive Health and Dysfunction, 2nd ed, pp. 89–124. Berlin: Springer-Verlag.
- Beutel ME, Weidner W, and Bra¨ hler E (2006) Epidemiology of sexual dysfunction in the male population. Andrologia 38: 115–121.

